Health
Doctor Answers Questions About COVID-19 Vaccination | Urgent Notice
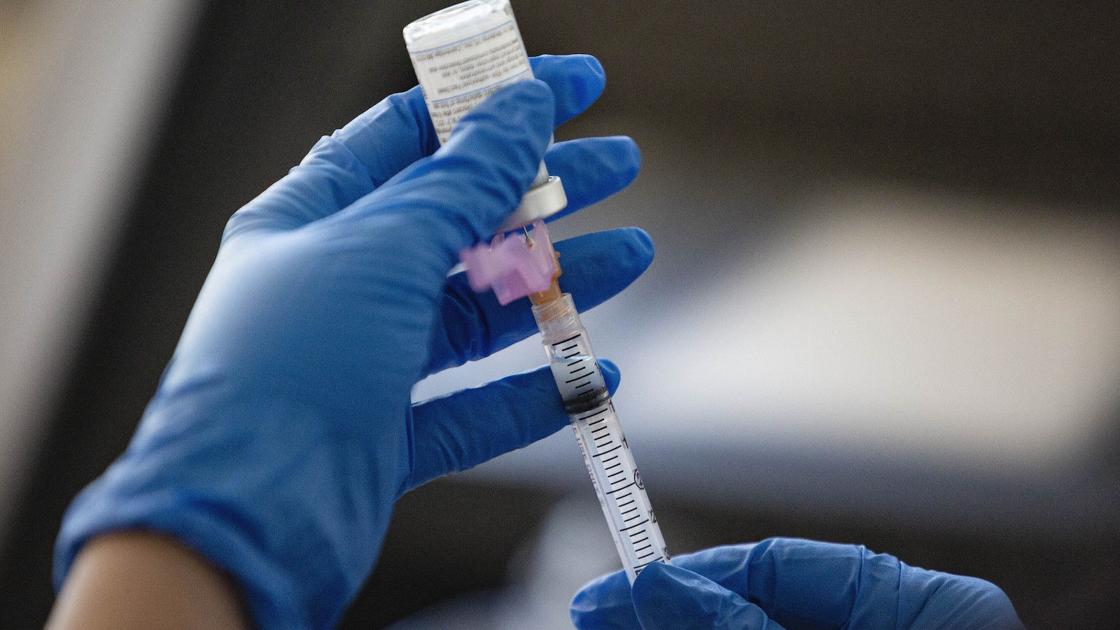

In early December, the Food and Drug Administration approved two COVID-19 vaccines for emergency use in the United States. The first is by Pfizer-BioNTech and the second is by Moderna. Both are recommended by the Centers for Disease Control and Prevention’s Immunization Implementation Advisory Board.
Vaccination in Maryland began on December 14, 2020. By Saturday, January 9, 123,971 Maryland residents had been vaccinated with the COVID-19 vaccine for the first time. 4,826 people were receiving a second dose.
As Marylanders and others throughout the United States continue to be vaccinated, there are many questions about these vaccines and vaccinations. Bay Times and Record Observer Interviewed with Dr. Eric Walgotz of Graysonville, Senior Staff Pathologist, Director of Medical Research Institute, Dean of Pathology, Professor Emeritus, Doctor’s Community Medical Center, Luminis Healthcare, Department of Pathology, George Washington University School of Medicine .. About vaccines.
“Both require two injections, the second injection being given three weeks after the first injection. After the second injection, clinical trials have shown that it is effective in preventing COVID-19 infection or severe infection. It was shown to be about 95%, “says Wargotz. “These clinical trials are underway to assess long-term effects, and additional COVID-19 vaccines are being tested.”
He said the COVID-19 vaccine from Oxford-Astra Zenaka also needed to be given twice and was recently approved in the UK and India.
Who will be vaccinated first?
Due to the limited supply of vaccines in the United States and the demand exceeding supply, the CDC has published guidelines on which groups should be prioritized. Each state is responsible for figuring out which citizens will be vaccinated first. Maryland has adopted the CDC recommendations.
“Vaccination is not mandatory. Currently, the first Phase 1A vaccinations are offered to front-line healthcare professionals and adults living and working in nursing homes, including nursing homes. Is an adult over the age of 75 and is a firefighter, police officer and other first responder, corrector, food and agricultural worker, US Postal Service worker, manufacturing worker, grocery store worker, public transport. Institutional workers and, according to Wargotz, people working in the education sector — teachers, support staff, day care workers.
“The third group, Phase 1C, includes people aged 65-74, people aged 16-64 and older with high-risk medical conditions, and other workers who are not considered to belong to Group 1b. It’s up to the individual states to decide. Those who fall into these second two categories: Transportation and Logistics, Food Services, Home Construction and Finance, Information Technology, Communications, Energy, Law, Media. May include people who work in public safety, and public health. “
People can contact their employer, online state health department, or local health department to see if they qualify as a member of any of these groups.
“ACIP closely monitors clinical trials in children and adolescents (aged 12+) and considers recommendations for use when the COVID-19 vaccine is approved for use in people under the age of 16.” Wargotz said. He was unaware of clinical trials in children under the age of 12.
“The goal is to provide everyone with COVID-19 vaccination as soon as sufficient vaccines are available. The CDC will vaccinate all US adults by the end of 2021. We expect to have enough vaccines for this, “said Wargotz.
“Traditional virus vaccines use dead or modified versions of the virus to stimulate the body and provoke an immune response, so they learn to recognize this virus and gain immunity.” Wargotz said.
“Pfizer and Moderna’s COVID-19 vaccine is an RNA vaccine. RNA is information inside the virus that allows the virus to generate all the viral mechanisms. These vaccines are extracted from the actual COVID-19 virus. It’s not made, it’s made in the lab, so it can be manufactured very quickly and brought to the test. When RNA enters the cells of your body, your cells have their own viral fragments. And it stimulates your body to gain immunity, “he explained. “These infused RNA itself is not harmful and the vaccine does not contain the complete virus, so these COVID-19 vaccines do not cause COVID-19 infection.”
The vaccine appears to be safe based on clinical trial information and weeks of experience with vaccines in the United States and other countries, he said.
“Serious reactions (via EpiPen) that require the use of EpiPen occur and are very rare, mainly due to irritants exposed by diet, breathing, contact, or injection in the past. It occurs in people who have experienced a serious allergic reaction that requires urgent treatment, “Walgotz said. “If you have any concerns about previous allergic reactions or allergies, please contact your healthcare provider before vaccination with COVID-19.”
The side effects of most vaccines occur within the first weeks or rarely months after vaccination. As a result, the FDA required companies to provide two months of safety data before applying for an emergency use authorization.
Information gathered in clinical trials, and more recently, side effects of the vaccine have been shown to include fever, headache, arm pain, redness at the injection site, and sometimes a feeling of weakness. He said these were also side effects noted with other viral vaccines.
Government regulators, healthcare organizations and manufacturers continue to collect safety data to understand long-term adverse effects, Wargotz added. He said clinical trials did not include women seeking pregnancy or pregnant women, so no information is available on the side effects of these groups.
Does the vaccine prevent COVID-19 infection?
Neither of the two approved vaccines is 100% effective in preventing infection, so some vaccinated people may be infected with COVID-19, he said.
“It is important to point out that vaccine trials have tested whether vaccines prevent viral infections in people. The trial focused on whether people are protected from the development of symptoms of the disease. Therefore, it is not clear whether vaccinated people can develop asymptomatic infections and spread the virus to others, “Wargotz said. “Some of the vaccinated people got sick with COVID-19, but they weren’t as sick as those who weren’t vaccinated.”
Some vaccines are better at reducing the severity of the disease than reducing or eliminating the infection.
Although the COVID-19 vaccine has some effect on the spread of the infection, it is unclear whether any of the approved vaccines is superior in controlling the infection and the severity of the disease.
“The 95% immunity achieved by people in the study required two injections of the vaccine every three weeks, so it is possible to get COVID-19 after the first injection.”
Do I still need to wear a mask and social distance after being vaccinated?
“The answer is simple-yes. The full protection provided by the vaccine will take at least a month to develop after the second injection,” Wargotz said.
Also, as mentioned earlier, it is not yet known whether the vaccine will prevent the infection or control the disease, so he said that the vaccinated person could be infected with the virus and infect others. Continued.
“Unfortunately, vaccinated people need to wear masks, stay socially distant, wash their hands and avoid large gatherings, especially indoors. This is on COVID-19. After infection, it will be necessary until a sufficient number of people have been vaccinated to obtain immunity or have achieved immunity until our health professionals determine that this number is epidemic / pandemic. Before declaring “over,” it is estimated to be 60-80% of the immunized population (so-called mass immunization), “he said.
Do I need to be vaccinated if I get COVID-19?
“It depends, but it’s probably,” Wargotz said. “It’s a good idea to have an antibody test to see if you’re already immune.”
If CDC is positive for COVID-19, the immune response will fluctuate, so healthcare professionals may recommend vaccination with COVID-19, and immunity does not last for more than 1 to 6 months. It states that there is a possibility. A recent study found that the innate immunity of COVID-19 was strong at 8 months. The CDC continues to evaluate the information to make specific timing recommendations.
What about immunity to new strains (mutants) of the virus?
“All viruses mutate. Viral genetics change in nature over time for a variety of reasons. The discovery of a more rapidly spreading strain of COVID-19 in the United Kingdom, Nigeria, and South Africa is a concern. “Wargotz said. “The identification of UK strains here in several states in the United States has heightened anxiety. The CDC suggests that approved vaccines are effective against these strains … The COVID-19 virus uses “peplomer” proteins to attach to lining cells of the respiratory tract. The COVID-19 variant has a slightly different composition of the “peplomer” protein, but the vaccine is still considered effective because there is still enough commonality between the original form and the variant. .. “
New UK stocks have been identified in four states in the United States. Not found in Maryland.
What should I expect before and after vaccination?
“If you are currently qualified or have to wait for the vaccine to become widely available, it is up to you to vaccinate. Highly recommended, but not required at this time,” Wargotz said. Mr. says.
“If you get the vaccine, you must receive a vaccine card or printout with the COVID-19 vaccine you received, the date you received it, and the place you received it. Also, the specific COVID you received or provided. -19 You will need to receive a paper or electronic version of the fact sheet detailing the vaccine. This fact sheet contains information that will help you understand the risks and benefits of receiving that particular vaccine. “
According to the protocol, all people vaccinated with COVID-19 should be monitored onsite for at least 15 minutes for no response.
The second vaccination must be from the same manufacturer.
“Overall, healthy adults should get it,” Walgotz recommended.
..
What Are The Main Benefits Of Comparing Car Insurance Quotes Online
LOS ANGELES, CA / ACCESSWIRE / June 24, 2020, / Compare-autoinsurance.Org has launched a new blog post that presents the main benefits of comparing multiple car insurance quotes. For more info and free online quotes, please visit https://compare-autoinsurance.Org/the-advantages-of-comparing-prices-with-car-insurance-quotes-online/ The modern society has numerous technological advantages. One important advantage is the speed at which information is sent and received. With the help of the internet, the shopping habits of many persons have drastically changed. The car insurance industry hasn't remained untouched by these changes. On the internet, drivers can compare insurance prices and find out which sellers have the best offers. View photos The advantages of comparing online car insurance quotes are the following: Online quotes can be obtained from anywhere and at any time. Unlike physical insurance agencies, websites don't have a specific schedule and they are available at any time. Drivers that have busy working schedules, can compare quotes from anywhere and at any time, even at midnight. Multiple choices. Almost all insurance providers, no matter if they are well-known brands or just local insurers, have an online presence. Online quotes will allow policyholders the chance to discover multiple insurance companies and check their prices. Drivers are no longer required to get quotes from just a few known insurance companies. Also, local and regional insurers can provide lower insurance rates for the same services. Accurate insurance estimates. Online quotes can only be accurate if the customers provide accurate and real info about their car models and driving history. Lying about past driving incidents can make the price estimates to be lower, but when dealing with an insurance company lying to them is useless. Usually, insurance companies will do research about a potential customer before granting him coverage. Online quotes can be sorted easily. Although drivers are recommended to not choose a policy just based on its price, drivers can easily sort quotes by insurance price. Using brokerage websites will allow drivers to get quotes from multiple insurers, thus making the comparison faster and easier. For additional info, money-saving tips, and free car insurance quotes, visit https://compare-autoinsurance.Org/ Compare-autoinsurance.Org is an online provider of life, home, health, and auto insurance quotes. This website is unique because it does not simply stick to one kind of insurance provider, but brings the clients the best deals from many different online insurance carriers. In this way, clients have access to offers from multiple carriers all in one place: this website. On this site, customers have access to quotes for insurance plans from various agencies, such as local or nationwide agencies, brand names insurance companies, etc. "Online quotes can easily help drivers obtain better car insurance deals. All they have to do is to complete an online form with accurate and real info, then compare prices", said Russell Rabichev, Marketing Director of Internet Marketing Company. CONTACT: Company Name: Internet Marketing CompanyPerson for contact Name: Gurgu CPhone Number: (818) 359-3898Email: [email protected]: https://compare-autoinsurance.Org/ SOURCE: Compare-autoinsurance.Org View source version on accesswire.Com:https://www.Accesswire.Com/595055/What-Are-The-Main-Benefits-Of-Comparing-Car-Insurance-Quotes-Online View photos
Pictures Credit
to request, modification Contact us at Here or [email protected]



